| Evoke little or no cellular inflammation (stealth). Cytopathic in vitro and in vivo. |
| Broad range of permissive cell types. |
| Fragmented, partial viral genome. |
| Molecularly heterogeneous. |
| Some are derived from simian cytomegaloviruses. |
| Vacuolated cytopathic effects in human and animal cell lines. |
| Herpesvirus-like particles seen on PCR generated products showed 50% relatedness to human cytomegalovirus. |
| Addtional sequencing showed 90 - 95% relatedness to African green monkey Simian Cytomegalovirus. |
| VIRUS | FASTA | BLASTN |
| Human CMV | 348 | 2.1 e-8 |
| Rhesus CMV | 1038 | 3.8 e-63 |
| Simian CMV | 2638 | 3.8 e-245 |
Memo
I told Dr. Murray that there was some concern at Lederle about a possible requirement barring the use of African green monkey kidney as teh substrate for the growth of attenuated polioviruses. Dr. Murray had stated that the adventitious agents that Dr. Kendall Smith is presumably detecting by his techniques are of little consequence for an oral preparation in that such a large experience exists with the use of oral polio vaccine without any evidence of trouble relating to these agents.
Lederle memo August 23, 1968
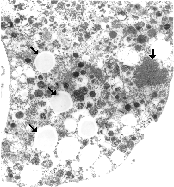
Electron micrograph of a vacuolated, "foamy cell." Arrows on the left indicate lipid-filled vacuoles. The amorphous grey mass indicated by the vertical arrow on the right is presumed to be a viral protein, the result of an unbalanced synthesis of viral components.

| Systemic infection |
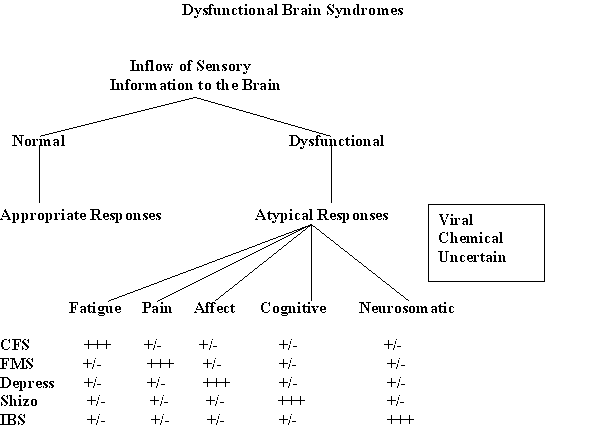
| Susceptibility of brain to localized infection. Wide range of dysfunctional brain diseases. |
| Subclinical dysfunction of many organs. |
| Leaky bowel, reduced liver detoxification can add a toxic encepalopathy component. |
| Virus induced tissue autoimmunity can induce endocrine damage, e.g. thyroid. |
| Illnesses among family members. |
| Illnesses in household pets. |
| Diverse clinical manifestations. |
| Probably antibody resistance to brain disease. |
| Oral, sexual and transplacental spread. |
| Stable virus, not easily inactivated. |
| Anti-viral treatments, gangcylovir, forscarnet, intereron. "Epione" an in vitro inhibitor. |
| Adjustments to sensory inputs, positive and negative influence. Placebo benefit. |
| Reduce exposure to toxic factors. |
| Support for detoxification pathways. |
| Specific therapies for complications e.g. endocrine dysfunction, secondary infections. |
| Culture virus from affected patients and controls. |
| Clone and sequence PCR amplified virus regions |
| Test family members for anti-viral antibodies. |
| Perform in-vitro anti-viral and anti-toxicity studies. |
| Undertake animal transmission studies. |
| Create treatment groups using current therapies. |